Featuring: Wake Forest® Baptist Health Steven Wong, Ph.D., DABCC (TC), FACB, Past President AACC, Professor of Pathology, serves as Director of the Clinical Chemistry and Toxicology Core Laboratory and is Co-director of the Clinical and Translational Mass Spectrometry...
Tags

Celebrating customer experience: Insights from SCIEX leaders
Introduction Customer Experience Day (CX Day) is a special occasion for SCIEX, celebrated every first Tuesday in October. It’s a day dedicated to recognizing the incredible value of our customers and the relentless dedication of our associates who strive to make...

LC-MS system replacement: Are you ready?
Meeting deadlines in a bioanalysis laboratory can be a big challenge. Older, less sensitive and less reliable LC-MS systems make it even more difficult. Even the disruption caused by the installation and validation can be disconcerting and delay decisions. Does this sound familiar?
Questions and answers to help improve your mycotoxin analysis
During a recent webinar I shared method details for mycotoxin analysis on the SCIEX 7500 system. In this blog i will share the Q&A for the submitted questions that we did not have chance to answer during the live webinar.
Optimized rolling collision energy curves for IDA and SWATH DIA for peptides
During data dependent acquisition (DDA or IDA) or SWATH acquisition, the collision energy can be automatically adjusted according to the mass/charge and charge of the peptide. This dependency has been well characterized on our QTOF systems. By selecting rolling...

Back to the new basics: Part 1 | Making the leap from GC-MS to LC-MS
Producing accurate results quickly in a demanding environment is no easy feat for analytical scientists. What’s more, many of us are constantly questioning ourselves—I certainly am—about whether we are employing the best technique for the analysis at hand.
It’s an overwhelming thought, considering the wide range of tools that are available to choose from, each of which offers varying levels of capacity, sensitivity, selectivity, specificity and cost. How do you meet the unique needs of your organization without breaking the bank? I get it, and I’m not here to convince you it’s easy. My aim is to guide you through the process to help you make the right decision for you.
MRM method transfer from a SCIEX Triple Quad or QTRAP 6500+ system to the SCIEX 7500 system
General recommendations when beginning method development Objective: The purpose of this document is to provide a quick reference for transferring MRM-based quantification methods from a SCIEX Triple Quad or QTRAP 6500+ system to a SCIEX 7500 system. While the best...
Adapting a SCIEX high flow source for microflow LC
To set up a SCIEX high flow source for microflow LC (Turbo V ion source, DuoSpray source or IonDrive Turbo V ion source), first you must replace the wider bore electrodes with more narrow bore hybrid electrodes. Note with the OptiFlow Turbo V ion sources, there are...

Identifying the unknown PFAS profile in firefighting foams/AFFF
According to a recent study from Harvard University, the US EPA, and NIEHS, traditional targeted analysis techniques poorly characterize the PFAS composition of contemporary PFAS-based firefighting foams, know as aqueous film-forming foams (AFFF). Using the EPA 533 PFAS drinking water method for the analyte list, the researchers found that targeted mass spectrometry methods accounted for <1% of organic fluorine content. This is important because it demonstrates that targeted analysis methods miss nearly all the PFAS compounds in modern AFFF mixtures, thus underestimating the risk to human health and the environment.
sMRM Concurrency Calculator
This excel sheet allows you to quickly estimate the MRM concurrency and approximate dwell time for your Scheduled MRM Algorithm acquisition method. Paste your MRM method with retention times into the input tab then calculate the Excel workbook. Plots will update to...

The risky business of aflatoxins in milk
If you’re in the dairy or food testing business, you know the threat aflatoxins pose. Aflatoxins are a type of mycotoxin produced by Aspergillus parasiticus, aspergillus flavus , and rarely aspergillus nomius.1 These are likely the most extensively researched group of mycotoxins because of their adverse health effects.2 What’s more, they are widely found in a variety of crops, namely maize, tree nuts, and spices. Believed to be primarily caused by rising temperatures and humidity, these naturally occurring fungi grow on crops in the field, or during storage of feed and raw materials, where they can potentially produce toxins that enter the food chain.

A Fleet of Analyzers Keeps Work Flowing
An Interview with Timothy Sangster, Head of Bioanalysis and Immunology, Charles River Laboratories, Edinburgh

Designed Specifically for Your Clinical Lab—Mass Spec Made Simple
Welcome to the first post in our clinical diagnostic blog series. Our ambition is to become your single destination for everything mass spec in the clinical diagnostic lab. To make this blog as useful as possible for you, we invite you to tell us what topics you would like us to cover. Please comment on this blog below and let us know what you’d like to hear!
Is Your Lab Prepared for Testing? The Global Supplement Market is Growing
Don’t judge a nutritional supplement by its label, as often, government monitoring of ingredients begins after the product enters the consumer market1. Meanwhile, there may be additional additives not mentioned on the label as they are used to address supplement side effects. Such is the case in the United States where even though federal law requires supplements to carry a dietary supplement label or a substitutional term, monitoring begins once a supplement is on the market. In China meanwhile, the China Food and Drug Administration’s (CFDA) health product potential illegal additives list, clearly stipulates monitoring processes for additives in six different types of nutritional supplements including weight loss, blood sugar reduction, blood pressure reduction, anti-fatigue, sleep improvement and immune strengthening functions.Read Tech Note >

How to Detect Additives in Cosmetics Amongst Ever Changing Regulations
In today’s technical blog, I’m talking about the cosmetics industry so let’s get right to it. According to a Research and Market report, “The Global Cosmetic market was $460 billion USD in 2014 and is estimated to reach 675 billion USD by 2020, growing at a rate of 6.4%.”1 The U.S. leads the pack with a reported $62 billion in revenue earned in 20162. So, what am I getting at? We know earnings are strong and consumers like their products. But the question remains, are these products that you put on your skin, hair, and ingest safe? Such is the thinking of scientists like me and other chemists who are routinely tasked with detecting minimal levels of potentially harmful ingredients in personal care products against ever-changing global regulations.

A Mine of Quantitative Proteomic Information
The Aebersold group at ETH Zurich focuses on proteomics research, including the development of techniques to study the proteome as an integrated entity. In collaboration with SCIEX, the group established SWATH® Acquisition mass spectrometry, a data-independent acquisition (DIA) method capable of fragmenting multiple peptide species concurrently. The resulting comprehensive data set can be retrospectively re-mined, enabling maximum benefit to be derived from any study.

Delivering New Biologics to the Marketplace
Characterization and quantification of host cell proteins (HCPs) in biopharmaceutical development and manufacturing is a critical step to ensuring product safety. While this can be achieved using ELISA, mass spectrometry using the SCIEX TripleTOF® 6600 System is more specific and enables the identification and quantitation of each of the individual proteins present.
Speeding the Development of Quantitative Biosimilar Assays
When developing new quantitative assays for Biotherapeutics, every biologic requires a specific sample prep strategy, which includes sourcing reagents and research protocols. However, as every bioanalytical lab knows all too well, it can also take up to two months to develop an optimized and robust LC-MS assay. For this reason, researchers understandably want an easier way to develop highly sensitive and specific assays for biotherapeutics and biosimilars to accelerate sample turnaround time.

The Benefits of Using SWATH Acquisition Technology when Testing Pesticides in Food
Up until recently, SWATH® Independent Data Acquisition (IDA), was not widely used for the detection of pesticides in food samples. Introduced in 2012, SWATH Acquisition is an advanced acquisition technology capable of running on high-resolution mass spectrometers such as the X500R QTOF system or Triple TOF technology. Originally used in the Omics market to ID and quantify complex samples, SWATH Acquisition is gradually making a transition across markets including the investigation of pesticides in food. Like designer drugs, pesticides continuously undergo synthesizing, and food labs are beginning to require a more reliable analysis method to be confident in their resulting reports.

Single Injection, Routine Antibiotic Testing in Urine Samples
The consumption of pharmaceuticals and personal care products is a day to day occurrence. Once consumed the body excretes the remaining part of the compound which is not absorbed. This waste, flushed down the toilet, makes its way through the sewage system before arriving at a treatment facility where it was then processed with chemicals to ensure its cleanliness. Despite being washed, there can remain trace amounts of bacteria, hormones, metals, and antibiotics in whatever you consume, not just water
Do You Want to Accelerate Quantitative Assays for Antibody Drug Conjugates?
Are you tasked with the bioanalysis of antibody drug conjugates (ADCs)? If so, you know they represent a rapidly growing class of biotherapeutics, but their unique chemical structure makes quantitative analysis particularly challenging.

Food Allergies – How Allergic Are You?
A recent study published by the Annals of Allergy, Asthma, and Immunology (ACAAI), pointed out, in a study of 109 people tested, that skin prick tests are not 100 percent reliable. In the study, participants were subjected to oral food challenges prior to skin testing in which 50 percent of individuals had no reaction. It was also discovered that blood tests were not full-proof even though they measure the presence of IgE antibodies to specific foods. These results are not surprising given that 50 to 60 percent of tests result in false-positives.
Comprehensive Therapeutic Protein Characterization Using One Single Method
Get to know how CESI-MS will allow you to quickly and accurately characterize protein therapeutics for attributes in a single method by downloading this discovery kit.

A Fresh Approach to Food Safety
The EU Reference Laboratory (EURL) for Fruits and Vegetables in Almería is responsible for a network of around 200 laboratories which provide essential surveillance and monitoring to ensure the safety of foodstuffs available across Europe. The EURL provides proficiency testing and method development for these official laboratories, ensuring rigorous screening to avoid harmful chemicals entering the food chain
The Only Solution You Need for Fast MetID
If you work in the breakneck world of therapeutic development, then you probably don’t even have time to read this blog (but we thought we would write it anyways, just in case). Drug metabolism samples are coming into your lab fast and furious. You need to turn them around in hours so that chemists and biologists can optimize the effectiveness of the therapeutic candidate. Time for lunch? We don’t think so!

The Key to Measuring Chemical Dyes in Food is LC-MS/MS
Adding colorful dyes to food is nothing new. In the early 19th century, for example, it wasn’t uncommon for manufacturers to add chalk to white bread, thicken milk with a lead compound, and inject red dye into meat in the quest for a fresher appearance1. Fast forward to the 21st century, however, and along with mass spectrometry, food standards have come a long way. Foods now must pass muster according to standards set by government regulators or else risk fines and punishment which can be costly for the manufacturer. To support these measures, are agencies such as the US-FDA, EFSA, and others which have banned some colors due to their toxic and carcinogenic nature which brings me to mass spectrometry analysis. Discover more when you read the following application note, “LC-MS/MS Analysis of Emerging Food Contaminants,” in which researchers used the ExionLC AD with a Phenomenex Column for sample separation followed by MS/MS detection with the SCIEX X500R QTOF system.

What is your Method for Separating Challenging Polar Molecules?
From small ions like phosphate, herbicide degradation to metabolites, oligosaccharides, peptides, and proteins. How is your lab analyzing polar molecules? The reason I ask is there is a saying, if you have a charged or polar molecule, look to capillary electrophoresis (CE) first. While liquid chromatography (LC) is an ideal front-end separation tool for many types of molecules, as the following poster points out, “From Small to Very Large, Orthogonal, Sensitive Polar Molecule Analysis by CESI-MS,” there are some situations that call for CE over LC analysis. For those of you that are not familiar with CESI-MS, it is the combining of CE separation with electrospray ionization, into one dynamic process, within the same device.

The Trouble with PFAS in Drinking Water
There has been a string of news articles concerning polyfluorinated alkyl substances (PFAS) in drinking water these days, and I must say, they have my attention. Here is the thing, when you think of drinking water in the United States, for example, crystal clear lakes, rivers, and groundwater along with effective water treatment come to mind. On the flip side, as safe, some supplies may be there are communities such as that of Flint, Michigan, which have been dealing with lead filled pipes for far too long. Contamination was so bad there that residents were provided bottled water for drinking purposes as the state decided who was responsible for replacing the affected water lines.
The Power Behind LC-MS for Quantifying mAb Therapeutics
Quantitation of monoclonal antibodies (mAbs) in biological fluids is important during all stages of antibody drug development. First developed in the 1970s, therapeutic mAbs have both research and medicinal impact as they can be used for diagnosis and treatment of a wide variety of diseases, and have a high level of specificity.
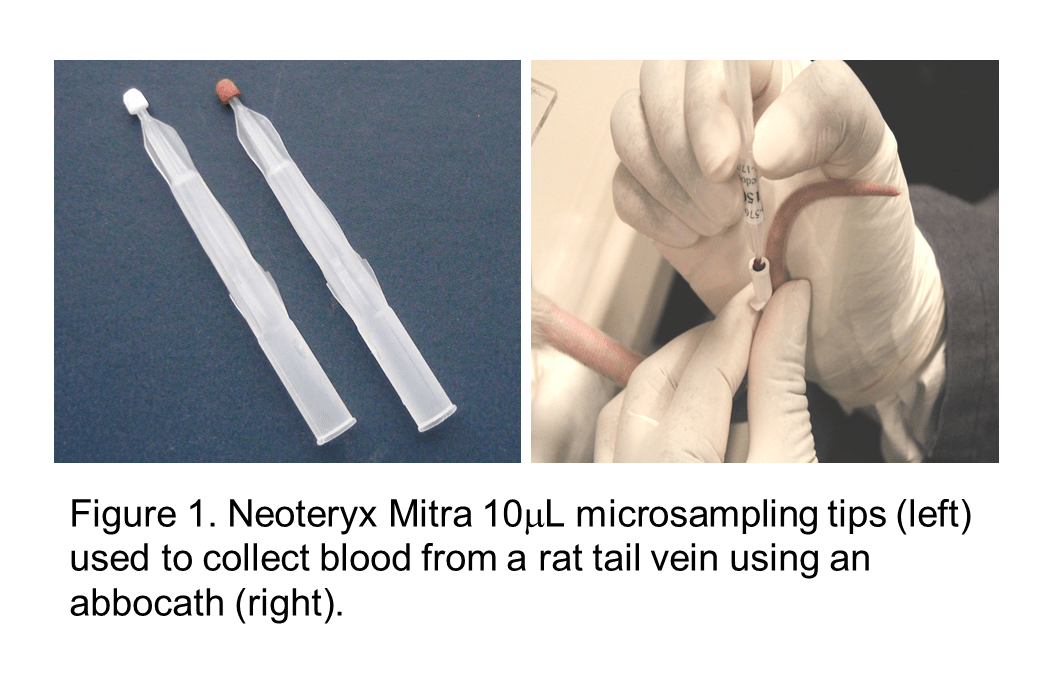
Volumetric Absorptive Microsampling and the SCIEX 6500+: A Pre-Clinical Case Study for the Biotherapeutic Exenatide
In an effort to Replace, Refine, and Reduce the number of animals used for pre-clinical research, several microsampling strategies have been implemented which allow for the consolidation of satellite TK and main study groups. In addition to the ethical gains driven by these 3Rs, microsampling has the potential of increasing scientific value since it becomes feasible to directly correlate exposure, toxicological effects and pharmacological response in the same individual

Say Goodbye to MetID Headaches, Say Hello to Automated Large Molecule Catabolism Processing
Is your mind swimming with large molecule catabolism data? Do you spend hours manually processing through complex spreadsheets to match your data with theoretical biologic catabolites? Are you wasting precious time by drawing out and processing your therapeutic peptide as a small molecule? Are you worried that you could be missing something?