We know that antibiotics used to treat livestock can end up in the food we eat. Routine food testing labs are essential for detecting compounds, like these, that can be dangerous to our health. Antibiotic residues include both parent molecules and metabolites left...
Tags
Overcoming uncertainty in your PFAS analysis
Just like gum on the bottom of a shoe, the existence of per- and poly-fluorinated alkyl substances (PFAS) in our environment is a sticky one. If you’re in the field of environmental testing, then you’re all too familiar with the threat these substances have on public health. While we have learned a lot about them over the years, there is still much more to understand. With the right detection methods, we can gather the information we need to empower us to make informed decisions on reducing the risks they impose.

6 Signs it’s time for a new vendor
A lab’s success depends on many factors from instrument quality to efficient operations, including being partnered with the right vendor. A vendor is more than just a supplier. They should provide you with a high-level quality of support in maximizing the lifespan and performance of your systems, reducing downtime, enhancing ROI and more. How do you know if you’re partnered with the right one? Here are six signs it might be time to find someone new.

Selecting an LC-MS system for quantitation of pharmaceutical drug development
We understand you are busy, needing to prioritize running instruments, reporting results and managing your laboratory to meet deadlines. We created a solution guide to explain how SCIEX systems fit in the drug development pipeline to save you time evaluating options.

Nitrosamines: Where are we now?
Nitrosamines are a large group of N-nitroso compounds that share a common functional N-N=O group. They are produced by a chemical reaction between a nitrosating agent and a secondary or tertiary amine. Back in 2018, nitrosamines suddenly found themselves in the spotlight when they were unexpectedly detected in medications for high blood pressure. Since then, they have been found in several other prescription medications, including those for heartburn, acid reflux and diabetes, resulting in manufacturers recalling some common medications.

Celebrating customer experience: Insights from SCIEX leaders
Introduction Customer Experience Day (CX Day) is a special occasion for SCIEX, celebrated every first Tuesday in October. It’s a day dedicated to recognizing the incredible value of our customers and the relentless dedication of our associates who strive to make...

PFAS analysis in food: a robustness study in sensitivity and stability
The combination of per- and polyfluoroalkyl substances (PFAS) testing, trace-level regulatory requirements and complex MS applications can be intimidating. In a recent webinar, now available on demand, SCIEX PFAS expert Craig Butt demonstrated how the new SCIEX 7500+ system can help make PFAS testing easier.

Your success and voice go a long way!
At the heart of everything we do is ensuring that your workflows and team are empowered to achieve optimal results with your SCIEX instruments, software, consumables, and services. Every interaction with SCIEX is designed to support your success through the dedication...

FDA’s final rule on LDTs: what does it mean for clinical laboratories?
On April 29, 2024, the U.S. Food and Drug Administration (FDA) announced a final rule regulating laboratory developed tests (LDTs) as in vitro diagnostic devices (IVDs) under the Federal Food, Drug and Cosmetic Act (FD&C Act). This rule amends FDA’s regulations to state that in vitro diagnostic tests “manufactured” by clinical laboratories fall within the scope of the FDA regulatory oversight and is poised to dramatically shift the way clinical diagnostic laboratories in the United States develop and offer LDTs in the future. Read this blog post for a basic overview of the scope, intent and implications of this final rule, including the regulatory requirements, exceptions and timeline for implementation.

LC-MS system replacement: Are you ready?
Meeting deadlines in a bioanalysis laboratory can be a big challenge. Older, less sensitive and less reliable LC-MS systems make it even more difficult. Even the disruption caused by the installation and validation can be disconcerting and delay decisions. Does this sound familiar?

An overview: LC-MS analysis of targeted protein degraders and their metabolites
Targeted protein degraders (TPD) are a relatively new therapeutic modality that opens the potential to target disease-causing proteins. These disease-causing proteins have been highly challenging for traditional small-molecule therapeutics to treat, making TPDs an exciting new therapeutic modality.

Is Your Beverage Truly 100% Fruit Juice?
There is nothing like the flavor of fruit juice whether freshly squeezed or made from concentrate to clench your thirst, except when it’s not 100 percent juice after all. As the following tech note, “Authenticity Assessment of Fruit Juices using LC-MS/MS and...
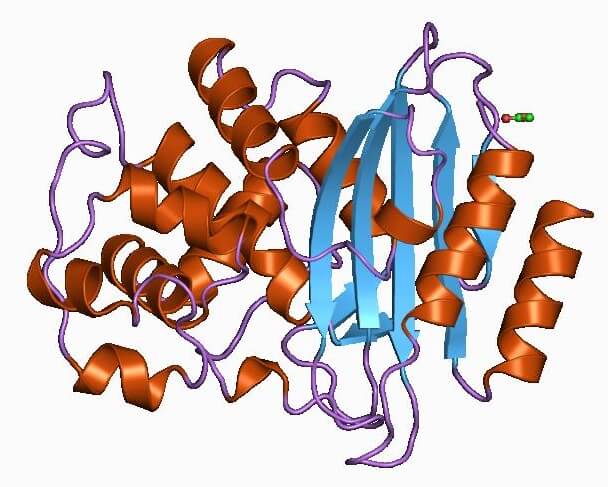
Superbugs, Antibiotic Resistance, and the QTRAP® 6500+ System
‘Superbugs’, or bacteria that have developed antibiotic resistance as a result of adapting to the drugs used in their treatment, are dangerous infections that doctors struggle to stop from spreading. Even common infections such as urinary tract infections and...
Vice President Biden Announces Agreement Naming Children’s Medical Research Institute’s ProCan Lab to the ‘Cancer Moonshot’ Initiative
A key goal of the ‘Cancer Moonshot’ initiative is the advancement of precision medicine, with the goal of making more targeted therapies available to more cancer patients. And researchers believe that the time is right, with the new technological innovations, the new insight into the biology of cancer and big improvements in the handling of ‘big data.’
It’s Time to Enhance Your Food Testing
Are you looking for ways to up the ante on your LC-MS/MS when it comes to food testing? Researchers here have developed a method for the analysis of approximately 400 pesticides in food samples, and their work is available for viewing in this year’s compendium.

Top Five Misconceptions about Mass Spectrometry
Do you work in a lab handling precious samples yet, hesitant to make the move to mass spectrometry? Many laboratories just like yours continue to conduct sample analysis using ELISA assays, PCR scans, and amino acid tests because of their effectiveness. These processes work, so why change? Well, these type of analytical experiments can report false positive and negative results. You have trained your staff, know the process, and fingers crossed, not too many user errors have compromised analysis.

Stoller Biomarker Discovery Centre, Addressing Some of the Biggest Issues in Medicine
The Stoller Biomarker Discovery Center, developed in partnership with SCIEX, was created to develop new omics technologies for biomarker research to understand the root cause of diseases such as cancer, cardiovascular disease, and autoimmune diseases. We initially announced our collaboration with the University of Manchester back in October 2015.

Rapid Separation Method for Intact Monoclonal Antibodies (Mab) Merges Charge Variant, Impurity, and Glycoform Analyses into a Single Assay
Throughout all stages of development and manufacture, monoclonal antibodies (mAbs) exhibit a great deal of structural complexity. After translation and folding, proteins undergo post-translational modifications, as well as spontaneous and enzymatic degradation, such that a single preparation of purified mAb exhibits a range of small structural changes, composed of various glycoforms and charge variants, as well as amino acids alterations due to oxidation, deamidation, isomerization, or other chemical reactions. This display of structural heterogeneity can influence the overall stability, efficacy, and safety profile; therefore, understanding the extent of structural modifications has become extremely important to drug manufacturers who continually assess mAb composition throughout bioprocessing to demonstrate stability, batch-to-batch consistency, and long-term shelf life.

Glycosylation Analysis Designed for the (Protein) Masses
A variety of post-translational modifications (PTMs) can impact a biotherapeutic protein’s mass, but none are as common as glycosylation.[1] Hence, the headline for a recent article in Genetic Engineering and Biotechnology News, “Post-Translational Icing on the Biologics Cake,” featuring comments from Sean McCarthy, Ph.D., Global Market Manager of Biologics at SCIEX.
The History of Isotopic Labels for Quantitative Proteomics
Proteomics has become a vital tool for biological scientists performing research on the healthy and diseased states of living things. It involves the large scale and systematic analysis of all proteins within a given cell, tissue, or organism. Because proteins are regulated by many different internal and external stimuli, the proteome is dynamic and quantities of proteins can change from one state to the next. Therefore, in order to be of the highest utility, proteomics experiments need to both identify and quantify proteins so that comparative studies can be done, such as between healthy cells and tumor cells, or the comparison of different treatment regimens.

Struggling to Analyze Small Volume Samples with Conventional LC-MS?
The M3 MicroLC System is designed for scientists who are struggling to analyze small volume samples with conventional LC-MS and need to lower their limits of quantitation while maintaining throughput and robustness. When designing the M3 MicroLC System, we...

Legal and Illicit Drugs in Wastewater Detected and Confirmed with QTRAP Technology
What happens when you up the sensitivity and lower detection limits on influent and effluent sewage tests? For starters, low levels of illegal drugs in samples begin to emerge. This is what researchers discovered when they combined the power of LC-MS/MS with the...
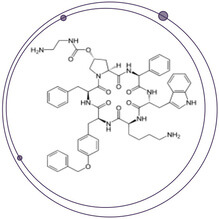
Using SelexION to Increase Selectivity for the Accurate and Sensitive Quantitation of a Difficult Peptide Therapeutic
SelexION® DMS Technology Drives Advancements in Challenging Large Molecule Bioanalysis By the End of 2024, the peptide therapeutics market value is expected to reach US$46.6 billion1. However, peptide therapeutics present some of the toughest analytical...
Harnessing the Power of MRM3 for Large Molecule Quantitative Bioanalysis
In a previous blog outlining the advantages of high-resolution accurate mass measurements for protein quantitation using the TripleTOF 6600, it was noted that although the triple-stage quadrupole demonstrated high sensitivity when operated in multiple reaction monitoring mode (MRM), the relatively low-resolution measurement of m/z failed to discriminate Rituximab response from nominally isobaric interferences given the complexity of the proteolytically digested samples (June 28/2016). While the accurate mass filtering capabilities of the TripleTOF 6600 represents one mechanism for achieving increased selectivity over MRM, the triple quadrupole/linear ion trap (LIT) hybrid platform represented by the QTRAP® 4500, 5500, 6500 and 6500+ systems provides an alternative technique by leveraging a third stage of MS, often referred to as MRM3. In this blog, we outline the MRM3 scan function and survey several large molecule applications which utilize the additional stage of fragmentation in the LIT to yield significant improvements in achievable detection limits when compared to MRM.

Taking care of your mass spectrometer—Onsite troubleshooting and maintenance training for today’s lab
Recently, we asked customers to tell us about their biggest challenges so we could customize training programs to meet the needs of today’s growing lab. Without hesitation, most of you said uptime and employee training are your most critical needs. As a result, our...

A Sting in the Tale for Neonicotinoids
Did you know one out of every three mouthfuls of your meal is a product of honeybee pollination—almonds and other tree nuts, berries, fruits, vegetables? To put numbers behind it, honeybee pollination amounts to about $15 billion of U.S....
The detection of acid herbicides and urons by large volume injection
Pre-treatment versus direct injection – that is the question posed in the application note, “The Detection of Acidic Herbicides and Phenyl Ureas by LC-MS/MS with Large Volume Injection and Automated Column Switching.” It’s just one of the dozens of articles you will find within the Environmental Compendium (pages 1 to 4, pesticides) now available for download.
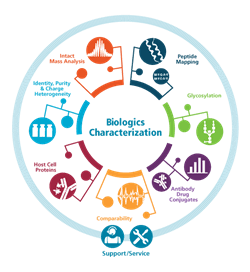
See What More You Can do With 360 Degree Biologics Characterization
Ever wish you had your own team of mass spectrometry experts at your side when working through biologics development and characterization challenges? With SCIEX 360° Innovations complex biologics characterization is streamlined with a full suite of mass spectrometry (MS), capillary electrophoresis (CE) systems, software, and services from SCIEX experts.
The Promise of Precision Medicine
Here is the latest update on the Worldwide Efforts to Accelerate Precision Medicine
The NIH recently issued a press release in early July announcing $55 million in awards. According to the release, the $55 million award in the fiscal year 2016 will go towards building the foundational partnerships and infrastructure needed to launch the Cohort Program of President Obama’s Precision Medicine Initiative (PMI). The PMI Cohort Program is a landmark longitudinal research effort that aims to engage 1 million or more U.S. participants to improve the ability to prevent and treat disease based on individual differences in lifestyle, environment, and genetics.

You’ve Seen It… Now Try It! BioPharmaView Software 2.0
At ASMS this year, the newest version of BioPharmaView Software was released. This software simplifies the processing of biotherapeutic data for characterization and comparability which can dramatically improve your productivity. BioPharmaView 2.0 Software accelerates characterization and comparability studies and simplifies reporting, so you can make better decisions, faster.

Quantitation of Antibiotics and Insecticides in Poultry Feed using LC-MS/MS
Quantitating antibiotics and insecticides in poultry is serious business. Overuse can lead to antibiotic resistance while insecticide residuals can cause harmful side effects in humans. In the United States, for example, the Federal Drug Administration (FDA), has offered up a plan to limit common antibiotics in feed, which are used to encourage growth. However, this is a voluntary plan, and as the following application note, “Quantitation of Antibiotics and Insecticides in Poultry Feed using LC-MS/MS,” points out, antibiotics have been shown to accumulate in poultry feathers, which are in turn used for nutritional elements in the feed.