Elevate Performance in Your Forensic Toxicology LabYou only need to skim through the United Nations World Drug Report 2018 to see that drug abuse is escalating. Associated with numerous medical, social, and legal problems, it comes as no surprise that ‘drugs of abuse’...
Tags

Celebrating customer experience: Insights from SCIEX leaders
Introduction Customer Experience Day (CX Day) is a special occasion for SCIEX, celebrated every first Tuesday in October. It’s a day dedicated to recognizing the incredible value of our customers and the relentless dedication of our associates who strive to make...

Detecting low levels of drugs and their metabolites in hair and nail samples using LC-MS/MS
You probably have heard of testing blood and urine samples for the presence of drugs and their metabolites. But do you know about the benefits of hair and nail analysis? In a recent webinar, Tina Binz, Deputy Head of the Center for Forensic Hair Analysis, University of Zurich, discussed the benefits of developing comprehensive and sensitive LC-MS/MS for the detection of low-level drugs and metabolites in keratinized matrices.

LC-MS system replacement: Are you ready?
Meeting deadlines in a bioanalysis laboratory can be a big challenge. Older, less sensitive and less reliable LC-MS systems make it even more difficult. Even the disruption caused by the installation and validation can be disconcerting and delay decisions. Does this sound familiar?
Questions and answers to help improve your mycotoxin analysis
During a recent webinar I shared method details for mycotoxin analysis on the SCIEX 7500 system. In this blog i will share the Q&A for the submitted questions that we did not have chance to answer during the live webinar.

Guide decisions during cell line development with more information at the intact level
Monitoring product quality attributes (PQAs) throughout monoclonal antibody (mAb) development is vital to ensuring drug safety and efficacy. By adopting orthogonal analytical techniques and integrating new technologies that have the potential to provide more information, it is possible to improve product quality and manufacturing efficiency and make more informed decisions.

Maximize NPS analysis with accurate mass spectrometry
LC-MS/MS is a powerful analytical tool in forensic toxicology testing that can support a variety of testing regimes such as screening, confirmation and quantitative workflows. More specifically, analysis of NPS using LC-MS/MS provides many advantages, including the ability to reliably detect new drugs and their metabolites from a variety of biological matrices.

Metabolite identification and peace of mind
Managing metabolite identification (Met ID) studies is challenging, so what is at the top of your priority list as you plan the year ahead? Ensuring you have the data needed to manage product safety, meeting deadlines, staff recruitment and training, maintaining compliance, capital expenses, or something else?

What has the Echo® MS system done for the pharma industry? (And don’t just take our word for it!)
SCIEX was very proud to have an illustration of the Acoustic Ejection Mass Spectrometry (AEMS) technology that powers the Echo® MS system on the front cover of the Journal of the American Society for Mass Spectrometry in January 2023. The associated article—Ultrahigh-Throughput Intact Protein Analysis with Acoustic Ejection Mass Spectrometry—was co-authored by scientists from SCIEX and Merck.
Methods for OPI electrode cleaning for Echo® MS system electrodes
Depending on the samples you are running on the system, it is possible for the Open Port Interface (OPI) electrode to become dirty or occluded over time. Below are two different cleaning strategies that can help you maintain your Echo® MS system and keep your OPI...

Rescheduling a Schedule I substance, and the Delta-8 controversy
Did you know that in the US, drugs and other chemicals are classified into 5 distinct categories depending on the drug’s acceptable medical use and its potential for abuse or dependency? Drugs federally classified as Schedule I substances by the US Drug Enforcement Administration (DEA) are considered to have the highest potential for abuse and for creating severe psychological and/or physical dependence. In addition to heroin, LSD and MDMA (ecstasy), cannabis is classified as a Schedule I substance in the Controlled Substance Act of 1970, which means it has no approved medical usage.

The Science Behind SelexION Differential Mobility Spectrometry Technology
Scientists and analysts across all fields of testing and research are increasingly challenged by complex samples requiring advanced analytical selectivity. And where LC-MS/MS sensitivity alone is not enough to meet the demands of modern day quantitative performance, Differential Ion Mobility Spectrometry (DMS) has proven to be a valuable addition.
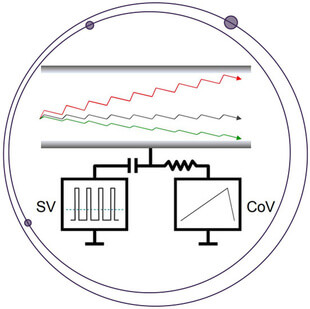
Elimination of Interference using the SelexION Differential Mobility System for the Quantitation of Rituximab in a Dual Surrogate Peptide Approach
The quantitation of proteins using the surrogate peptide approach can complicate nominal mass Triple Quadrupole MRM measurements due to co-extracted interference when using non-selective extraction techniques such as pellet digestion. High resolution coupled with accurate mass filtering can mitigate such interference, as reported previously for the determination of rituximab using the TripleTOF 6600 (Protein Quant Approaches). However, an additional level of selectivity can often be achieved on nominal mass systems using the orthogonal gas-phase separation approach offered by the SelexION+® Differential Mobility System technology (DMS). Interfaced between the sampling orifice and ion source, the DMS separates ions based upon differences in their migration rates under alternating low and high field waveform amplitudes (Figure 1). Ion clustering in low fields and declustering in high fields amplifies the distinction in mobility of an ion, resulting in improved resolution from interfering species of differing molecular cross-section.1-4

Happy Birthday to SWATH Acquisition! 5 Years of Innovation
With its introduction at the HUPO World Congress in 2010 in Sydney Australia by Ruedi Aebersold, SWATH® Acquisition instantly intrigued scientists around the world. Here was a new technique with the potential to revolutionize the way proteomics studies were performed! Based on a data independent acquisition strategy using a SCIEX TripleTOF® 5600 system, SWATH was able to consistently identify and quantify at least as many peptides and proteins as other far more mature proteomics strategies on the market, but with quantitative accuracy and reproducibility rivaling gold standard MRM experiments! This solution was made broadly available to researchers with a full launch of SWATH Acquisition in the Analyst® TF 1.6 Software on the TripleTOF 5600+ System at ASMS 2012 in Vancouver (A Mine of Quantitative Proteomic Information. Prof Dr. Ruedi Aebersold, Head of the Department of Biology, ETH Zurich).

5 Tips for Calibrating a QTOF Mass Spectrometer
Do you have questions about your mass spec? How about a workflow? Our community members are involved in active discussions and receive expert answers from customers like you, SCIEX scientists, and support specialists every week. One recent topic concerned the automatic calibration on TripleTOF® systems as answered by Dr. Christie Hunter whose focus is developing and testing innovative MS workflows for omics research through working collaboratively with the instrument, chemistry, and software research groups.

Enhancing In Vitro ADME Screening
LC-MS technology is helping contract research organization Cyprotex Discovery Ltd. perform bioanalysis of small molecules, peptides, and other pharmaceuticals, enabling quantification to be performed in complex matrices during in vitro ADME studies.
Host Cell Protein Analysis – Mass Spec’s Edge Over ELISA
The number of protein based drugs coming onto the market is at an all-time high, particularly those produced with a host cell system. With host cells come their own proteins. These host cell proteins (HCPs) constitute a major part of process-related impurities and can adversely affect drug safety, so it is critical that they are identified and quantified accurately.
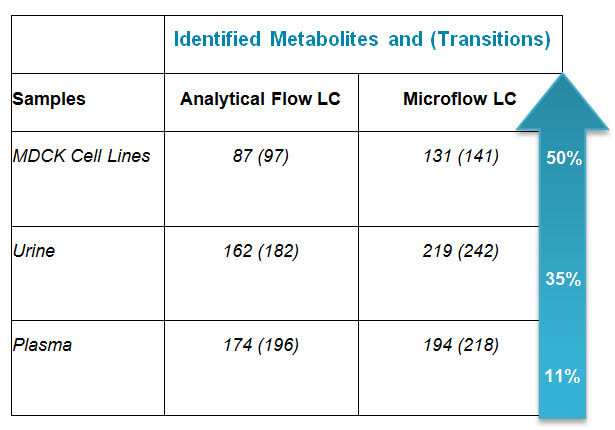
Why Microflow HILIC Chromatography for Targeted Metabolomics Applications?
I recently had the opportunity to catch up with Baljit Ubhi to discuss the top questions you’re asking in regards to using Microflow HILIIC Chromatography for Targeted Metabolomics. Here’s what Bal said:

Fipronil Tainted Eggs Detected in Several European Countries
News agencies all over the world are reporting a new food contamination issue regarding eggs which have been found to contain residues of Fipronil. According to Nieuwsuur, a Dutch news, and current affairs program, “The Fipronil scandal is a huge blow to the poultry sector. Millions of eggs are destroyed and 138 companies remain tentatively closed. But supermarkets also face great damage. In recent days all contaminated eggs have been taken out of the shelves.” CBS news has reported that contaminated eggs have been discovered in Belgium and in the Netherlands with other European countries now on alert.

In Search of the Unknown
The production of high-quality drinking water entails rigorous treatment and testing procedures. For water suppliers’ laboratories, such as the Zweckverband Landeswasserversorgung in Germany, one of the major challenges is the identification of trace levels of organic substances, which can be achieved with the help of mass spectrometry.

Discover The Benefits of Knowledge Base Articles
Did you know you can access Knowledge Base Articles for trending user questions compiled and answered by SCIEX support experts? Doing so may help to reduce your support calls, not to mention downtime. Instead of waiting for a problem to occur, you can stay on top of it, and be a part of the solution. To give you an idea of trending articles, consider the how this past month saw questions and answers including:

A Fleet of Analyzers Keeps Work Flowing
An Interview with Timothy Sangster, Head of Bioanalysis and Immunology, Charles River Laboratories, Edinburgh

Designed Specifically for Your Clinical Lab—Mass Spec Made Simple
Welcome to the first post in our clinical diagnostic blog series. Our ambition is to become your single destination for everything mass spec in the clinical diagnostic lab. To make this blog as useful as possible for you, we invite you to tell us what topics you would like us to cover. Please comment on this blog below and let us know what you’d like to hear!
Is Your Lab Prepared for Testing? The Global Supplement Market is Growing
Don’t judge a nutritional supplement by its label, as often, government monitoring of ingredients begins after the product enters the consumer market1. Meanwhile, there may be additional additives not mentioned on the label as they are used to address supplement side effects. Such is the case in the United States where even though federal law requires supplements to carry a dietary supplement label or a substitutional term, monitoring begins once a supplement is on the market. In China meanwhile, the China Food and Drug Administration’s (CFDA) health product potential illegal additives list, clearly stipulates monitoring processes for additives in six different types of nutritional supplements including weight loss, blood sugar reduction, blood pressure reduction, anti-fatigue, sleep improvement and immune strengthening functions.Read Tech Note >

How to Detect Additives in Cosmetics Amongst Ever Changing Regulations
In today’s technical blog, I’m talking about the cosmetics industry so let’s get right to it. According to a Research and Market report, “The Global Cosmetic market was $460 billion USD in 2014 and is estimated to reach 675 billion USD by 2020, growing at a rate of 6.4%.”1 The U.S. leads the pack with a reported $62 billion in revenue earned in 20162. So, what am I getting at? We know earnings are strong and consumers like their products. But the question remains, are these products that you put on your skin, hair, and ingest safe? Such is the thinking of scientists like me and other chemists who are routinely tasked with detecting minimal levels of potentially harmful ingredients in personal care products against ever-changing global regulations.

A Mine of Quantitative Proteomic Information
The Aebersold group at ETH Zurich focuses on proteomics research, including the development of techniques to study the proteome as an integrated entity. In collaboration with SCIEX, the group established SWATH® Acquisition mass spectrometry, a data-independent acquisition (DIA) method capable of fragmenting multiple peptide species concurrently. The resulting comprehensive data set can be retrospectively re-mined, enabling maximum benefit to be derived from any study.

Delivering New Biologics to the Marketplace
Characterization and quantification of host cell proteins (HCPs) in biopharmaceutical development and manufacturing is a critical step to ensuring product safety. While this can be achieved using ELISA, mass spectrometry using the SCIEX TripleTOF® 6600 System is more specific and enables the identification and quantitation of each of the individual proteins present.
Speeding the Development of Quantitative Biosimilar Assays
When developing new quantitative assays for Biotherapeutics, every biologic requires a specific sample prep strategy, which includes sourcing reagents and research protocols. However, as every bioanalytical lab knows all too well, it can also take up to two months to develop an optimized and robust LC-MS assay. For this reason, researchers understandably want an easier way to develop highly sensitive and specific assays for biotherapeutics and biosimilars to accelerate sample turnaround time.

The Benefits of Using SWATH Acquisition Technology when Testing Pesticides in Food
Up until recently, SWATH® Independent Data Acquisition (IDA), was not widely used for the detection of pesticides in food samples. Introduced in 2012, SWATH Acquisition is an advanced acquisition technology capable of running on high-resolution mass spectrometers such as the X500R QTOF system or Triple TOF technology. Originally used in the Omics market to ID and quantify complex samples, SWATH Acquisition is gradually making a transition across markets including the investigation of pesticides in food. Like designer drugs, pesticides continuously undergo synthesizing, and food labs are beginning to require a more reliable analysis method to be confident in their resulting reports.

Single Injection, Routine Antibiotic Testing in Urine Samples
The consumption of pharmaceuticals and personal care products is a day to day occurrence. Once consumed the body excretes the remaining part of the compound which is not absorbed. This waste, flushed down the toilet, makes its way through the sewage system before arriving at a treatment facility where it was then processed with chemicals to ensure its cleanliness. Despite being washed, there can remain trace amounts of bacteria, hormones, metals, and antibiotics in whatever you consume, not just water
Do You Want to Accelerate Quantitative Assays for Antibody Drug Conjugates?
Are you tasked with the bioanalysis of antibody drug conjugates (ADCs)? If so, you know they represent a rapidly growing class of biotherapeutics, but their unique chemical structure makes quantitative analysis particularly challenging.