Craig Butt explains a non-targeted omics approach to characterizing and profiling compounds in citrus oil Read time: 4 minutes There is increasing interest among consumers in the benefits of natural products containing citrus beyond the traditionally known benefits of...
Tags
Pharma perspectives: The influence of LC-MS innovation on drug development outsourcing
It is no secret that (bio)pharmaceutical research and development is complex, both scientific and regulatory processes. Working for a contract research organization and more recently for SCIEX has provided an interesting perspective on trends the market experiences that affect many of us.
Overcoming uncertainty in your PFAS analysis
Just like gum on the bottom of a shoe, the existence of per- and poly-fluorinated alkyl substances (PFAS) in our environment is a sticky one. If you’re in the field of environmental testing, then you’re all too familiar with the threat these substances have on public health. While we have learned a lot about them over the years, there is still much more to understand. With the right detection methods, we can gather the information we need to empower us to make informed decisions on reducing the risks they impose.
6 Signs it’s time for a new vendor
A lab’s success depends on many factors from instrument quality to efficient operations, including being partnered with the right vendor. A vendor is more than just a supplier. They should provide you with a high-level quality of support in maximizing the lifespan and performance of your systems, reducing downtime, enhancing ROI and more. How do you know if you’re partnered with the right one? Here are six signs it might be time to find someone new.

Selecting an LC-MS system for quantitation of pharmaceutical drug development
We understand you are busy, needing to prioritize running instruments, reporting results and managing your laboratory to meet deadlines. We created a solution guide to explain how SCIEX systems fit in the drug development pipeline to save you time evaluating options.

Nitrosamines: Where are we now?
Nitrosamines are a large group of N-nitroso compounds that share a common functional N-N=O group. They are produced by a chemical reaction between a nitrosating agent and a secondary or tertiary amine. Back in 2018, nitrosamines suddenly found themselves in the spotlight when they were unexpectedly detected in medications for high blood pressure. Since then, they have been found in several other prescription medications, including those for heartburn, acid reflux and diabetes, resulting in manufacturers recalling some common medications.

PFAS analysis in food: a robustness study in sensitivity and stability
The combination of per- and polyfluoroalkyl substances (PFAS) testing, trace-level regulatory requirements and complex MS applications can be intimidating. In a recent webinar, now available on demand, SCIEX PFAS expert Craig Butt demonstrated how the new SCIEX 7500+ system can help make PFAS testing easier.

Your success and voice go a long way!
At the heart of everything we do is ensuring that your workflows and team are empowered to achieve optimal results with your SCIEX instruments, software, consumables, and services. Every interaction with SCIEX is designed to support your success through the dedication...

FDA’s final rule on LDTs: what does it mean for clinical laboratories?
On April 29, 2024, the U.S. Food and Drug Administration (FDA) announced a final rule regulating laboratory developed tests (LDTs) as in vitro diagnostic devices (IVDs) under the Federal Food, Drug and Cosmetic Act (FD&C Act). This rule amends FDA’s regulations to state that in vitro diagnostic tests “manufactured” by clinical laboratories fall within the scope of the FDA regulatory oversight and is poised to dramatically shift the way clinical diagnostic laboratories in the United States develop and offer LDTs in the future. Read this blog post for a basic overview of the scope, intent and implications of this final rule, including the regulatory requirements, exceptions and timeline for implementation.

LC-MS system replacement: Are you ready?
Meeting deadlines in a bioanalysis laboratory can be a big challenge. Older, less sensitive and less reliable LC-MS systems make it even more difficult. Even the disruption caused by the installation and validation can be disconcerting and delay decisions. Does this sound familiar?

An overview: LC-MS analysis of targeted protein degraders and their metabolites
Targeted protein degraders (TPD) are a relatively new therapeutic modality that opens the potential to target disease-causing proteins. These disease-causing proteins have been highly challenging for traditional small-molecule therapeutics to treat, making TPDs an exciting new therapeutic modality.

Stop Fumbling Around in the Dark: Try LC-MS/MS for Designer Drugs in Forensic Samples
Imagine being blindfolded, sent into a large (dark) room filled with obstacles and challenged to find an item, but you don’t know what that item is, and you have never seen it before. Then you must do the same again the next day, but you are looking for a different...

Something’s in the Water: Tackle Your PFAS Analysis
Exposure to per- and polyfluoroalkyl substances (PFAS) residues could be dangerous. These chemicals have been linked to a variety of adverse health effects including liver damage, thyroid disease, decreased fertility, high cholesterol, obesity, hormone suppression,...

5 Reasons Why Mass Spec is Transforming Clinical Labs
According to the American Clinical Laboratory Association, more than 7 billion clinical lab tests are performed in the U.S. every year. While mass spectrometry represents only a fraction of the clinical applications, there are a growing number of tests where mass...

Save Effort. Save Time. Consolidate Your BioTherapeutic Quality, Safety, and Efficacy Assessments with One Direct LC-MS MAM Assay.
In a previous blog, we learned how you could Simplify Your Life with a Streamlined Workflow for Multiple Attribute Methodology (MAM). Based on the well-known technique of peptide mapping, a Liquid Chromatography-Mass Spectrometry-based MAM workflow (LC-MS MAM) enables...

Metabolism and Biotransformation – What Are You Missing?
Are you looking to improve your MetID studies? Want to achieve the productivity and throughput you need to keep your projects moving and have complete confidence that you’re not missing any low level or parent-like metabolites in your sample? In this blog, we’re...

Biologics Characterization: Intact and Subunit Mass Analysis
When All You Need Is Range – Mass Range, Dynamic Range, and the Complete Range Launching the best possible product in the shortest time possible is key for pharmaceutical companies like you. As you know, nearly every process throughout the biologics development cycle...

Now You Can Overcome Your Food and Environmental Testing Obstacles
How do you safeguard human health and protect your reputation through meeting stringent global food and environmental safety standards? When it comes to testing food and environmental samples, the quality of your testing data is vital to your business. You need fast...

Unraveling the Metabolome
Dr. David Wishart, Departments of Biological Sciences and Computing Science, University of AlbertaCanada’s TMIC is an internationally-recognized leader in metabolomics, providing tools and resources that are used by laboratories around the world. The center offers...

It’s Time to Stop Fraudulent Food Reaching the Consumer with LC-MS/MS
If you are reading this blog, it is likely that you are fully acquainted with food fraud, but let’s just take a moment to set the scene. Food fraud is by no means a new phenomenon and goes as far back as the middle ages, with the concept of tampering or...

Product Quality Assessments of Biotherapeutics Using LC-MS MAM
Why LC-MS MAM?The Liquid Chromatography-Mass Spectrometry Multiple Attribute Methodology (LC-MS MAM) is a technique that is quickly gaining traction in the development and manufacturing of biopharmaceuticals. But what exactly is MAM? As researchers describe in a...
Ion Formation Control Is the Key to Increasing System Robustness
Traditional mass spectrometry ionization methods such as electronic spray ionization (ESI) and matrix-assisted laser desorption ionization (MALDI), are popular methods for detecting the molecular weight of proteins, peptides, and other biologics. The reason being both...

Why You Need Mass Spec for Veterinary Drug Residue and Antibiotic Analysis
As hospitals struggle with increasingly hard-to-treat antimicrobial resistance (ARM) bacterial infections, related deaths now exceed 700,000 per year globally and are predicted to reach 10 million per year by 2050. There’s no denying that these statistics are both...

Simplify Your Life with a Streamlined Workflow for Multiple Attribute Methodology (MAM)
The effort to fully characterize and release a biotherapeutic to the market can be onerous. Typically, many tests are required to identify and monitor various attributes of the final product in order to ensure the safety and efficacy of the drug. These product quality...

Solving the Mystery of Forensic Quantitative Analysis
Why You Need Differential Mobility Spectrometry for Forensic Mass Spec Analysis Forensics depend on detection of even the smallest compounds to deliver results you can rely on. You need fast analysis methods that provide highly accurate data across a multitude of...

Calling all Bioanalysts: We’re Making Bioanalytical Selectivity Challenges History
High selectivity is a key component of successful quantitative bioanalysis. As a bioanalyst, we need consistently accurate and robust quantitation of small molecule therapeutics and metabolites. Challenged by complicated matrix interferences, high baseline signal, and...

The Latest and Greatest for Improving Lipidomic Analysis by Mass Spectrometry
Differential Mobility Separation (DMS) resolves multiple lipid classes within complex lipid matrices prior to MS analysis to enable more confident identification of lipid species and more accurate quantitation by MS/MS. Lipidomics research has progressed rapidly in...

Out with the “If it Ain’t Broke, Don’t Fix it” Approach. In with Remote Monitoring.
Often by the time you’re aware of a problem, it’s too late, and disaster has already struck. You’re forced to pick up the pieces—downtime, reduced productivity and lost data. When your PC goes down, IT comes to the rescue. That’s a reactive approach. In many cases, IT...

Increase your Lab’s ROI with Remote Monitoring Service for SCIEX LC-MS Systems
Like most organizations, your lab is likely thinking about ways to reduce costs and maximize profits.If so, keep in mind that no matter your area of research, sample volume or locations, remote monitoring services can make a difference in yearly maintenance...
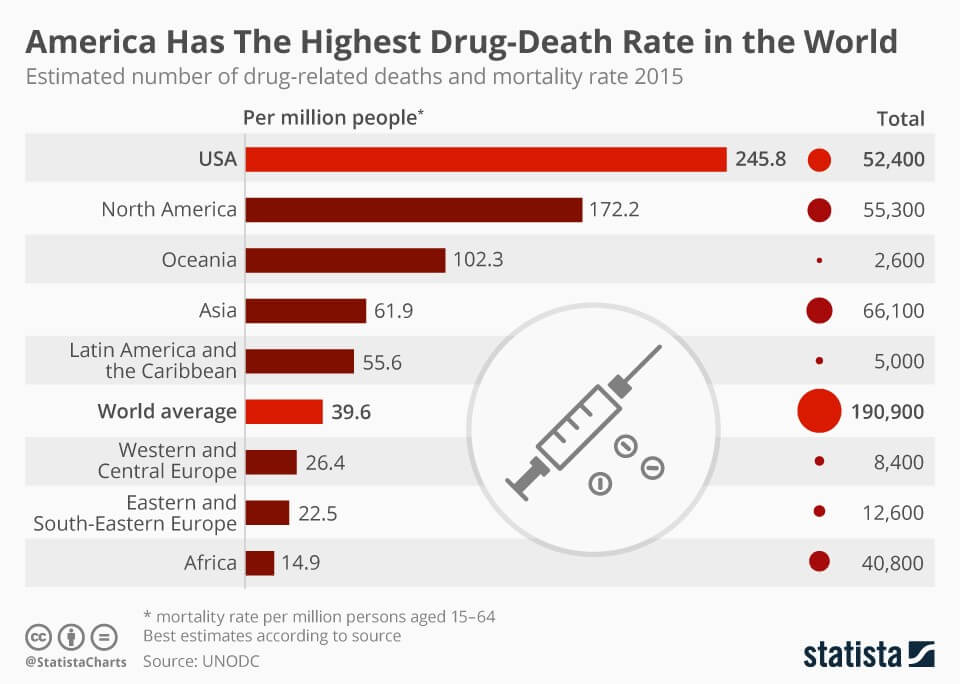
SWATH Acquisition: A Step Closer to Unravelling the Opioid Puzzle
Drug overdose has become one of the leading causes of death of Americans under 50. If that’s not bad enough, let’s put this into perspective. America makes up only 4% of the world population. In 2015, it recorded 52,400 drug-related deaths, which is about 27% of the...

Metabolomics Studies Benefit Biomedical Research
Professor Dr. Thomas Hankemeier, Head of the Division of Systems Biomedicine and Pharmacology, LACDRLACDR is a center of excellence for multidisciplinary research into drug discovery and development, with a strong focus on metabolomics. As part of its research...